Sato, Fernando; Braga, Scheila F; Santos, Helio F dos; Galvao, Douglas S
Structure-Activity Relationship Investigation of Some New Tetracyclines by Electronic Index Methodology Journal Article
In: arXiv preprint arXiv:0708.2931, 2007.
@article{sato2007structure,
title = {Structure-Activity Relationship Investigation of Some New Tetracyclines by Electronic Index Methodology},
author = {Sato, Fernando and Braga, Scheila F and Santos, Helio F dos and Galvao, Douglas S},
url = {http://arxiv.org/abs/0708.2931},
year = {2007},
date = {2007-01-01},
journal = {arXiv preprint arXiv:0708.2931},
abstract = {Tetracyclines are an old class of molecules that constitute a broad-spectrum antibiotics. Since the first member of tetracycline family were isolated, the clinical importance of these compounds as therapeutic and prophylactic agents against a wide range of infections has stimulated efforts to define their mode of action as inhibitors of bacterial reproduction. We used three SAR methodologies for the analysis of biological activity of a set of 104 tetracycline compounds. Our calculation were carried out using the semi-empirical Austin Method One (AM1) and Parametric Method 3 (PM3). Electronic Indices Methodology (EIM), Principal Component Analysis (PCA) and Artificial Neural Networks (ANN) were applied to the classification of 14 old and 90 new proposed derivatives of tetracyclines. Our results make evident the importance of EIM descriptors in pattern recognition and also show that the EIM can be effectively used to predict the biological activity of Tetracyclines.},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
Braga, Scheila Furtado; Galvao, Douglas Soares
Benzo [c] quinolizin-3-ones theoretical investigation: SAR analysis and application to nontested compounds Journal Article
In: Journal of chemical information and computer sciences, vol. 44, no. 6, pp. 1987–1997, 2004.
@article{braga2004benzo,
title = {Benzo [c] quinolizin-3-ones theoretical investigation: SAR analysis and application to nontested compounds},
author = {Braga, Scheila Furtado and Galvao, Douglas Soares},
url = {http://pubs.acs.org/doi/abs/10.1021/ci049837u},
year = {2004},
date = {2004-01-01},
journal = {Journal of chemical information and computer sciences},
volume = {44},
number = {6},
pages = {1987--1997},
publisher = {American Chemical Society},
abstract = {We investigate with the use of theoretical methodologies the activity of a set of 41 benzo[c]quinolizin-3-
ones (BC3), some of them explored as selective inhibitors of the human 5R-reductase steroid. For the
structure-activity study we have considered dividing the molecules into groups of tested and nontested
compounds. Semiempirical calculations and pattern recognition methods such as Electronic Indices
Methodology (EIM), Principal Components Analysis (PCA), Hierarchical Cluster Analysis (HCA), and
K-Nearest Neighbors (KNN) have been applied to search for a correlation between experimental activity
and theoretical descriptors. Our results show that it is possible to directly correlate some molecular quantum
descriptors with BC3 biological activity. This information can be used in principle to identify active/inactive
untested compounds and/or to design new active compounds.},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
ones (BC3), some of them explored as selective inhibitors of the human 5R-reductase steroid. For the
structure-activity study we have considered dividing the molecules into groups of tested and nontested
compounds. Semiempirical calculations and pattern recognition methods such as Electronic Indices
Methodology (EIM), Principal Components Analysis (PCA), Hierarchical Cluster Analysis (HCA), and
K-Nearest Neighbors (KNN) have been applied to search for a correlation between experimental activity
and theoretical descriptors. Our results show that it is possible to directly correlate some molecular quantum
descriptors with BC3 biological activity. This information can be used in principle to identify active/inactive
untested compounds and/or to design new active compounds.
Coluci, Vitor Rafael; Vendrame, Rosana; Braga, RS; Galvao, DS
In: Journal of chemical information and computer sciences, vol. 42, no. 6, pp. 1479–1489, 2002.
@article{coluci2002identifying,
title = {Identifying relevant molecular descriptors related to carcinogenic activity of polycyclic aromatic hydrocarbons (PAHs) using pattern recognition methods},
author = {Coluci, Vitor Rafael and Vendrame, Rosana and Braga, RS and Galvao, DS},
url = {http://pubs.acs.org/doi/abs/10.1021/ci025577%2B},
year = {2002},
date = {2002-01-01},
journal = {Journal of chemical information and computer sciences},
volume = {42},
number = {6},
pages = {1479--1489},
publisher = {American Chemical Society},
abstract = {Polycyclic Aromatic Hydrocarbons (PAHs) constitute an important family of molecules capable of inducing
chemical carcinogenesis. In this work we report structure-activity relationship (SAR) studies for 81 PAHs
using the pattern-recognition methods Principal Component Analysis (PCA), Hierarchical Clustering Analysis
(HCA) and Neural Networks (NN). The used molecular descriptors were obtained from the semiempirical
Parametric Method 3 (PM3) calculations. We have developed a new procedure that is capable of identifying
the PAHs’ carcinogenic activity with an accuracy higher than 80%. PCA selected molecular descriptors
that can be directly correlated with some models proposed to PAHs’ metabolic activation mechanism leading
to the formation of PAHs-DNA adducts. PCA, HCA and NN validate the energy separation between the
highest occupied molecular orbital and its next lower level as a major descriptor defining the carcinogenic
activity. This descriptor has been only recently discussed in the literature as one new possible universal
parameter for defining the biological activity of several classes of compounds.},
keywords = {},
pubstate = {published},
tppubtype = {article}
}
chemical carcinogenesis. In this work we report structure-activity relationship (SAR) studies for 81 PAHs
using the pattern-recognition methods Principal Component Analysis (PCA), Hierarchical Clustering Analysis
(HCA) and Neural Networks (NN). The used molecular descriptors were obtained from the semiempirical
Parametric Method 3 (PM3) calculations. We have developed a new procedure that is capable of identifying
the PAHs’ carcinogenic activity with an accuracy higher than 80%. PCA selected molecular descriptors
that can be directly correlated with some models proposed to PAHs’ metabolic activation mechanism leading
to the formation of PAHs-DNA adducts. PCA, HCA and NN validate the energy separation between the
highest occupied molecular orbital and its next lower level as a major descriptor defining the carcinogenic
activity. This descriptor has been only recently discussed in the literature as one new possible universal
parameter for defining the biological activity of several classes of compounds.
2007
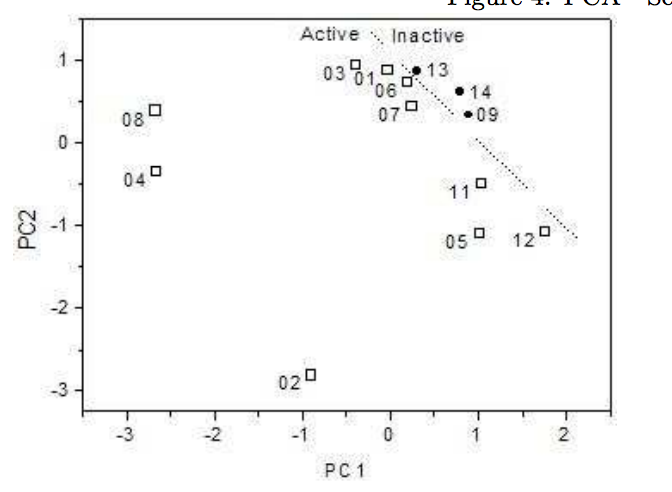
Sato, Fernando; Braga, Scheila F; Santos, Helio F dos; Galvao, Douglas S
Structure-Activity Relationship Investigation of Some New Tetracyclines by Electronic Index Methodology Journal Article
In: arXiv preprint arXiv:0708.2931, 2007.
Abstract | Links | BibTeX | Tags: Drug Design, Electronic Structure, Neural Networks, PCA/HCA, Tetracyclines, Theory of Electronic Indices
@article{sato2007structure,
title = {Structure-Activity Relationship Investigation of Some New Tetracyclines by Electronic Index Methodology},
author = {Sato, Fernando and Braga, Scheila F and Santos, Helio F dos and Galvao, Douglas S},
url = {http://arxiv.org/abs/0708.2931},
year = {2007},
date = {2007-01-01},
journal = {arXiv preprint arXiv:0708.2931},
abstract = {Tetracyclines are an old class of molecules that constitute a broad-spectrum antibiotics. Since the first member of tetracycline family were isolated, the clinical importance of these compounds as therapeutic and prophylactic agents against a wide range of infections has stimulated efforts to define their mode of action as inhibitors of bacterial reproduction. We used three SAR methodologies for the analysis of biological activity of a set of 104 tetracycline compounds. Our calculation were carried out using the semi-empirical Austin Method One (AM1) and Parametric Method 3 (PM3). Electronic Indices Methodology (EIM), Principal Component Analysis (PCA) and Artificial Neural Networks (ANN) were applied to the classification of 14 old and 90 new proposed derivatives of tetracyclines. Our results make evident the importance of EIM descriptors in pattern recognition and also show that the EIM can be effectively used to predict the biological activity of Tetracyclines.},
keywords = {Drug Design, Electronic Structure, Neural Networks, PCA/HCA, Tetracyclines, Theory of Electronic Indices},
pubstate = {published},
tppubtype = {article}
}
2004
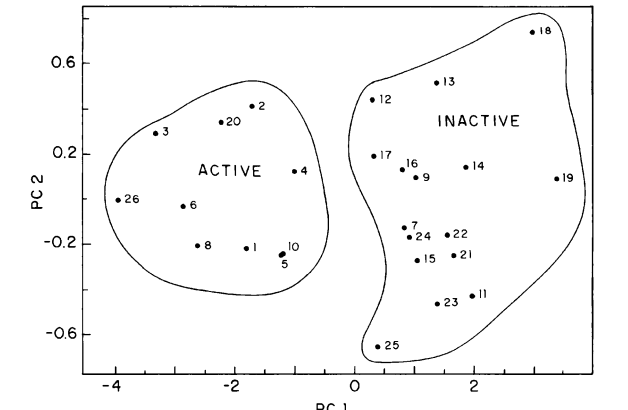
![Benzo [c] quinolizin-3-ones theoretical investigation: SAR analysis and application to nontested compounds](https://sites.ifi.unicamp.br/galvao/files/2015/02/Screen-Shot-2015-02-26-at-12.21.40-PM.png)
Braga, Scheila Furtado; Galvao, Douglas Soares
Benzo [c] quinolizin-3-ones theoretical investigation: SAR analysis and application to nontested compounds Journal Article
In: Journal of chemical information and computer sciences, vol. 44, no. 6, pp. 1987–1997, 2004.
Abstract | Links | BibTeX | Tags: Drug Design, Electronic Structure, PCA/HCA, Theory of Electronic Indices
@article{braga2004benzo,
title = {Benzo [c] quinolizin-3-ones theoretical investigation: SAR analysis and application to nontested compounds},
author = {Braga, Scheila Furtado and Galvao, Douglas Soares},
url = {http://pubs.acs.org/doi/abs/10.1021/ci049837u},
year = {2004},
date = {2004-01-01},
journal = {Journal of chemical information and computer sciences},
volume = {44},
number = {6},
pages = {1987--1997},
publisher = {American Chemical Society},
abstract = {We investigate with the use of theoretical methodologies the activity of a set of 41 benzo[c]quinolizin-3-
ones (BC3), some of them explored as selective inhibitors of the human 5R-reductase steroid. For the
structure-activity study we have considered dividing the molecules into groups of tested and nontested
compounds. Semiempirical calculations and pattern recognition methods such as Electronic Indices
Methodology (EIM), Principal Components Analysis (PCA), Hierarchical Cluster Analysis (HCA), and
K-Nearest Neighbors (KNN) have been applied to search for a correlation between experimental activity
and theoretical descriptors. Our results show that it is possible to directly correlate some molecular quantum
descriptors with BC3 biological activity. This information can be used in principle to identify active/inactive
untested compounds and/or to design new active compounds.},
keywords = {Drug Design, Electronic Structure, PCA/HCA, Theory of Electronic Indices},
pubstate = {published},
tppubtype = {article}
}
ones (BC3), some of them explored as selective inhibitors of the human 5R-reductase steroid. For the
structure-activity study we have considered dividing the molecules into groups of tested and nontested
compounds. Semiempirical calculations and pattern recognition methods such as Electronic Indices
Methodology (EIM), Principal Components Analysis (PCA), Hierarchical Cluster Analysis (HCA), and
K-Nearest Neighbors (KNN) have been applied to search for a correlation between experimental activity
and theoretical descriptors. Our results show that it is possible to directly correlate some molecular quantum
descriptors with BC3 biological activity. This information can be used in principle to identify active/inactive
untested compounds and/or to design new active compounds.
2002
Coluci, Vitor Rafael; Vendrame, Rosana; Braga, RS; Galvao, DS
In: Journal of chemical information and computer sciences, vol. 42, no. 6, pp. 1479–1489, 2002.
Abstract | Links | BibTeX | Tags: Carcinogenesis, Neural Networks, PCA/HCA, Polycyclic Aromatic Hydrocarbons (PAHs), Theory of Electronic Indices
@article{coluci2002identifying,
title = {Identifying relevant molecular descriptors related to carcinogenic activity of polycyclic aromatic hydrocarbons (PAHs) using pattern recognition methods},
author = {Coluci, Vitor Rafael and Vendrame, Rosana and Braga, RS and Galvao, DS},
url = {http://pubs.acs.org/doi/abs/10.1021/ci025577%2B},
year = {2002},
date = {2002-01-01},
journal = {Journal of chemical information and computer sciences},
volume = {42},
number = {6},
pages = {1479--1489},
publisher = {American Chemical Society},
abstract = {Polycyclic Aromatic Hydrocarbons (PAHs) constitute an important family of molecules capable of inducing
chemical carcinogenesis. In this work we report structure-activity relationship (SAR) studies for 81 PAHs
using the pattern-recognition methods Principal Component Analysis (PCA), Hierarchical Clustering Analysis
(HCA) and Neural Networks (NN). The used molecular descriptors were obtained from the semiempirical
Parametric Method 3 (PM3) calculations. We have developed a new procedure that is capable of identifying
the PAHs’ carcinogenic activity with an accuracy higher than 80%. PCA selected molecular descriptors
that can be directly correlated with some models proposed to PAHs’ metabolic activation mechanism leading
to the formation of PAHs-DNA adducts. PCA, HCA and NN validate the energy separation between the
highest occupied molecular orbital and its next lower level as a major descriptor defining the carcinogenic
activity. This descriptor has been only recently discussed in the literature as one new possible universal
parameter for defining the biological activity of several classes of compounds.},
keywords = {Carcinogenesis, Neural Networks, PCA/HCA, Polycyclic Aromatic Hydrocarbons (PAHs), Theory of Electronic Indices},
pubstate = {published},
tppubtype = {article}
}
chemical carcinogenesis. In this work we report structure-activity relationship (SAR) studies for 81 PAHs
using the pattern-recognition methods Principal Component Analysis (PCA), Hierarchical Clustering Analysis
(HCA) and Neural Networks (NN). The used molecular descriptors were obtained from the semiempirical
Parametric Method 3 (PM3) calculations. We have developed a new procedure that is capable of identifying
the PAHs’ carcinogenic activity with an accuracy higher than 80%. PCA selected molecular descriptors
that can be directly correlated with some models proposed to PAHs’ metabolic activation mechanism leading
to the formation of PAHs-DNA adducts. PCA, HCA and NN validate the energy separation between the
highest occupied molecular orbital and its next lower level as a major descriptor defining the carcinogenic
activity. This descriptor has been only recently discussed in the literature as one new possible universal
parameter for defining the biological activity of several classes of compounds.
http://scholar.google.com/citations?hl=en&user=95SvbM8AAAAJ


